Overview
Over the last decades, cannabis plants (Cannabis sativa L.) have received significant academic and pharmaceutical attention for their physiological, disease-fighting and quality-of-life improving properties. Most of this attention has centred around the understanding of one class of biologically active ingredients: the cannabinoids.
These compounds were first discovered in the 1940s and the structure of the most known cannabinoid (Δ9-tetrahydrocannabinol (“THC”)) was determined in 1964 in Israel. As of 2020, more than 100 different cannabinoids have been identified from Cannabis sativa L. and new cannabinoids continue to be discovered, for example cannabidiphorol (CBDP) en Δ9-tetrahydrocannabiphorol (Δ9-THCP) in 2019. The two most well-known and studied compounds originating from the cannabis plant are Δ9-tetrahydrocannabinol (“THC”) and cannabidiol (“CBD”).
However, many of the other cannabinoids, routinely described as minor cannabinoids since they are not produced in large quantities in cannabis plants, have also demonstrated unique pharmacological properties and mechanisms apart from those of THC and CBD. Each individual cannabinoid can affect a range of different receptors in the human body, including, but not limited to, the endocannabinoid receptors.
In this way, they are responsible for a wide variety of pharmacological effects. Limited preclinical and clinical research have resulted in the lack of a full understanding of the pharmacological effects of cannabinoids (and possible combinations thereof).
Cannabinoids
In general, the term cannabinoids refer to a group of chemical substances, regardless of their origin or structure, that bind to specific cannabinoid receptors (CB receptors) in the human body. They are a large and varied group of molecules that can be classified in several different ways, but the most useful way to understand the cannabinoid diversity is the following:
- Phytocannabinoids: plant-derived cannabinoids such as those that can be isolated from the cannabis plant.
- Endogenous cannabinoids (“endocannabinoids”): produced inside the mammalian body
- Synthetic cannabinoids: produced in the laboratory.
Phytocannabinoids are plant-derived cannabinoids produced by glandular trichomes covering the surface of the cannabis flowers (and sometimes the leaves). They are found exclusively in cannabis plants although some other plants also produce very small amounts of cannabinoid(-like) compounds, such as the Echinacea species, Helichrysum umbraculigerum and Spilanthes oleracea.
These phytocannabinoids are often extracted from plant biomass using both solvent (e.g. ethanol) and solvent-less extraction methods (supercritical CO2 extraction) and are purified to obtain up to 98% pure molecules.
By 2020, more than 100 different phytocannabinoids have been identified, of which cannabidiol (CBD) and Δ⁹-Tetrahydrocannabinol (THC) are the best known and studied. However, the pharmacological potential of other lesser-known cannabinoids (e.g. Cannabigerol (CBG), Tetrahydrocannabivarin (THCV), etc.) is starting to attract more attention from the scientific community.
Endogenous cannabinoids, or endocannabinoids, are cannabinoids produced inside the mammalian body and function as lipid-based retrograde neurotransmitters.
These compounds were discovered in the mid ‘90s along with the specific cannabinoid receptors: (a) CB1R primarily located in the brain and central nervous system; (b) CB2R, mainly localized in the peripheral organs and the immune system. The discovery of these elements also led to the development of the term “the Endocannabinoid System (ECS)”.
The two best known and studied endocannabinoids are anandamide (N-arachidonoylethanolamine) and 2-arachidonoylglycerol (2-AG). Endocannabinoids and by extension the endocannabinoid system are involved in a wide variety of processes, including pain, memory, mood, appetite, stress, sleep, metabolism, immune function, and reproductive function. Endocannabinoids are arguably one of the most widespread and versatile signalling molecules known to man.
The main difference between phytocannabinoids, endocannabinoids and synthetic cannabinoids is that the latter are fully synthetic and created in the laboratory. These synthetic cannabinoids can be:
(a) completely structurally analogous with phytocannabinoids, for example dronabinol (Δ9-THC synthetic), which is the active compound of MARINOL®, a medicine that comes in capsules and has been consumed in the US since 1985 to prevent nausea, vomiting, loss of appetite and loss of weight
(b) New Chemical Entities (NCEs) such as nabilone, the active substance of CESAMET®, a medicine approved for nausea and vomiting control caused by cancer chemotherapy.
These synthetic cannabinoids can be produced in one of two ways:
- Chemical synthesis: Compounds are man-made in a laboratory. This process leads to ultra-pure compounds, which makes it easy to develop reliable, safe, stable and easy-to-dose cannabinoid medicine.
- Biosynthesis: In this process, micro-organisms such as yeast or bacteria are used to produce exact copies of phytocannabinoids.
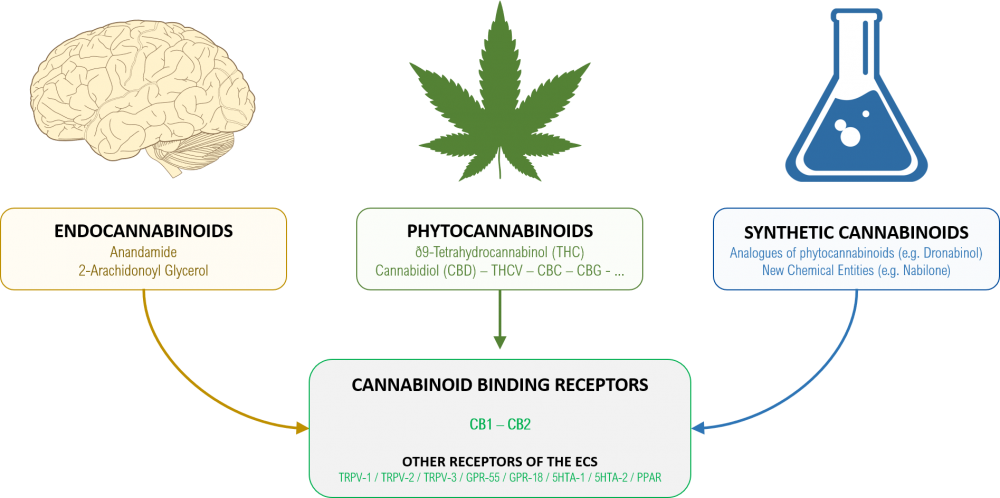
Image: Cannabinoids can have different origins. But they still bind to the same receptor network.

Cannabinoid receptor type 1 (CB1) is expressed in the peripheral nervous system and central nervous system (brain), but also in the lungs, liver and kidneys. CB1-receptors are thought to be one of the most widely expressed Gαi protein-coupled receptors in the brain and are encoded by the CNR1 gene in humans. The CB1 receptor can be activated by endocannabinoids (anandamide is the primary endogenous agonist), several phytocannabinoids (such as Δ9-THC) and synthetic analogs of cannabinoids.
Cannabinoid receptor type 2 (CB2) is expressed mainly on T cells of the immune system, on macrophages and B cells, and in hematopoietic cells. They also have a function in keratinocytes. They are also expressed on peripheral nerve terminals. In the brain, they are mainly expressed by microglial cells, where their role remains unclear. CB2-receptors are encoded by the CNR2 gene in humans. The CB2 receptor can be activated by endocannabinoids (2-Arachidonoylglycerol is the primary endogenous agonist), several phytocannabinoids (such as Δ9-THC, THCV) and synthetic analogs of cannabinoids.
Cannabinoid Receptors
The ability of cannabis plant extracts (Cannabis sativa L.) to produce various medicinal effects was recognized as early as the third millennium B.C. when Chinese texts described its usefulness in the relief of pain and cramps.
The highly lipophilic nature of the biologically active ingredients of cannabis (cannabinoids) led to the idea that cannabis triggered its effects in a nonspecific way by disrupting the membrane lipids of cells. However, the discovery of cannabinoid receptors in the 1980s in the mammalian body completely changed this way of thinking. The existence of cannabinoid receptors in the brain was discovered from in vitro studies, with the receptor designated as the cannabinoid receptor type 1 or CB1. In 1993 a second brain cannabinoid receptor named cannabinoid receptor type 2 or CB2 was identified.
Cannabinoid receptors are of a class of cell membrane receptors in the G protein-coupled receptor superfamily. As is typical of G protein-coupled receptors, the cannabinoid receptors contain seven transmembrane spanning domains. There are currently two known subtypes of cannabinoid receptors, termed CB1 and CB2.
The last decade has also seen mounting evidence for the existence of alternative receptors/ion channels that can be activated by cannabinoids collectively called “non-CB1/CB2 receptors” such as GPR18; GRP55; GPR119; serotonin 1A receptor (5-TH1A); Thermo transient receptor potential channels (Thermo-TRPVs such as TRPV1, TRPV2, TRPA1, TRPM8) and perixosome proliferator-activated receptors (PPAR).
Industry concepts of cannabinoid-based medicines
Medicinal cannabis, cannabinoid-based medicine,… what’s in a name?
This infographic provides an overview of the various concepts.
Cannovex is focusing on concept 4.0 – i.e. the development of cannabinoid-based medicines.

Our Partners
Cannovex BV
BE0656.491.644
Agoralaan Abis
3590 Diepenbeek
Belgium

© 2020 – Cannovex BV – All rights reserved
Tel: +32 11 28 69 00
E-mail: info@www.cannovex.com




